Q. How do you remove biofilm? (The featured image is sourced from here.)
A. In order to “kill off” any microbial residue, a cleaning and removal of inanimate and organic matter is a must. This would follow with the disinfecting, “kill” step if you will, with an EPA registered disinfectant.
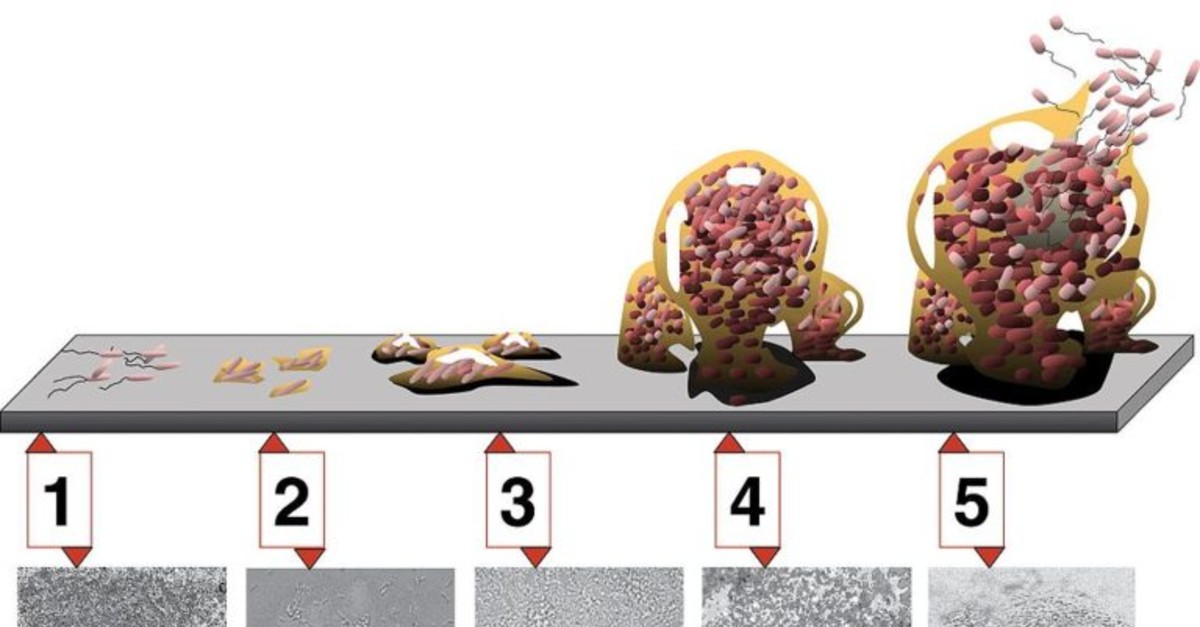
Biofilm typically consists of bacteria adhered to a surface, forming a complex architecture of microbial communities. An established biofilm has a matrix composed of microbial cells and cell-secreted extracellular polymeric substance (EPS) typically involving hydrophobic polysaccharides. This matrix forms a defined architecture and provides an optimal environment for microbes to communally evolve to adapt to their environment. Biofilms can thus be tenacious and difficult to eliminate.
When removing biofilm, both low-foaming clean-in-place (CIP) as well as higher-foaming clean-out-of-place (COP) enzymatic detergents are effective.
- Low-foaming CIP detergents: A reliable way to remove biofilms from stainless steel process piping and tank surfaces is to use a two-step, high-temperature process with an aqueous alkaline cleaner (Solujet® Low-Foaming Phosphate-Free Liquid), followed by an aqueous acidic cleaner (Citrajet® Low-Foam Liquid Acid Cleaner/Rinse)
For heavy biofilm applications or for biofilms that have persisted over a long period of time, a typical effective process consists of a 3% alkaline cleaner at 75°C applied for 30 minutes, followed by a cursory rinse to mitigate neutralizing the next acid step. The second step uses a 3% acidic cleaner at 75°C applied for 30 minutes, followed by a final thorough rinse with water. This two-step process, at the specified temperatures, produces a very clean result on stainless steel. It can be desirable to use a disinfectant after cleaning to slow down the return of the biofilm.
These methods may be used to clean and prepare tanks, process lines, centrifuges, and similar equipment. In addition to removing well-established biofilms, biofilm cleaning processes are typically incorporated into preventive maintenance programs to prevent biofilm formation.
- High-foaming manual / COP detergents: When using scrubbing, soaking, sonication, or recirculation applications that avoid high pressure/air entrainment (e.g., filter cleaning), use an enzymatic cleaner that contains protease, surfactants, and builders (e.g. Tergazyme® Enzyme-Active Powdered Detergent)
Such formulations remove biofilm at lower temperatures than typically used in CIP systems. Low temperatures are needed to avoid denaturing the enzymes used for cleaning. The protease addresses both cells and proteins bound in the lipopolysaccharide portion of the biofilm, whereas the surfactants improve wetting and penetration through the hydrophobic lipopolysaccharide portion of the biofilm. When used appropriately, this solution completely removes the inanimate portions of biofilm.
To manually clean or remove biofilm, use Tergazyme detergent at 3% in warm water (35°C–55°C to ensure optimal enzyme activity). These detergents are high-foaming cleaners and are not suitable for spray-in-air CIP systems. Solutions can be pumped and gently agitated; however, using high agitation at an air/solution interface will result in excessive foam. For an old biofilm, consider a 30-minute 3% detergent solution (soak/recirculation) at 35°C before the acid/alkaline cleaning protocol above.
This approach with enzymatic detergent also can be used reliably in manual surface cleaning and cleaning preparation for subsequent disinfectant-based biofilm removal, such as in cleanrooms. Removing proteinaceous, organic, and inanimate residues of biofilm will lengthen the amount of time before the biofilm returns. Use enzymatic cleaning processes in intermittent preventive maintenance between alkaline/acidic cleaning to remove dead cells and traces of lipopolysaccharide portions of biofilm.
Bleach alone is sometimes used to treat biofilms, but often the hydrophobic nature of the biofilm interferes with the efficacy of oxidization; therefore, bleach alone may not be sufficient. Incorporating an alkaline cleaner or detergent improves the effectiveness of biofilm removal compared to cleaning with bleach alone. Bleach used at concentrations suitable for food contact surfaces does have some efficacy on thermophilic bacilli and similar biofilms, although efficacy may be intermittent. The bleaching mechanism breaks down membranes at sulfhydryl groups and unsaturated side chains; as potential exposed sites are used up, efficacy of the cleaning may fail, allowing the biofilm to persist. Further, minimizing use of bleach may be preferable in light of concerns regarding the potential for bleach to cause chloride stress cracking on stainless steel.
Biofilms can be extremely tough. We are always pleased to discuss your particular application further. Contact us any time!
To request these or any Alconox, LLC detergent for free, please complete the questionnaire at Get Sample. For more information about any one of our Alconox, LLC detergents, consult the technical bulletin for each product. Or click here to access each of our detergent’s Safety Data Sheets.
Do you have a critical cleaning question for the experts at Alconox, LLC? Search TechNotes to see if it’s been answered before or Ask Alconox.




