Q: What is the importance of a Critical Micelle Concentration of a detergent and why is it important for critical cleaning?
A: Thank you for your question about the Critical Micelle Concentration (CMC) of surfactants in Alconox, LLC detergents.
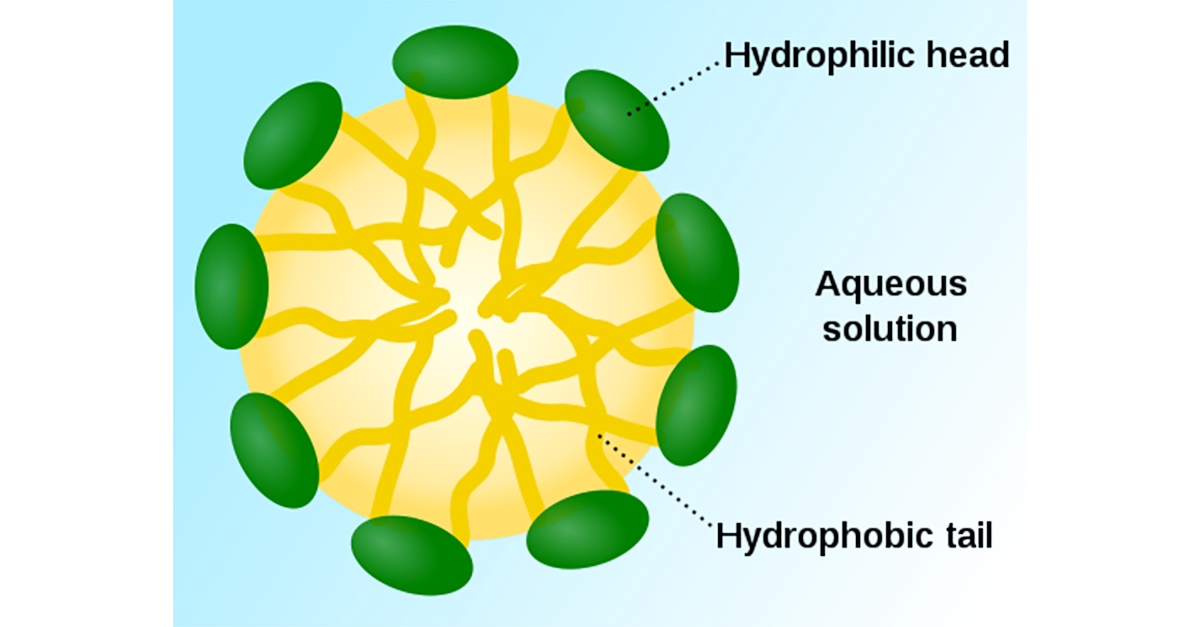
The critical micelle concentration is the minimum concentration at which the emulsifiers in a detergent will coalesce in to membrane structures of globes, rods or sheets with their hydrophobic (water hating) ends on the inside of the membrane and their hydrophilic (water loving) ends on the outside such that hydrophobic oily residues can be emulsified inside these membrane structures. Micelles are further elaborated on below. If you are below the critical micelle concentration, you do not have effective emulsifying of oily residues.

Alconox, LLC detergents are typically recommended for use at 4 times higher concentration than the theoretical CMC to ensure there are available micelles and cleaning capacity, as well as the ability to form new micelles as micelles are filled up with emulsified oils. Note that the addition of residues and particularly electrolytes can alter the CMC, so in real world applications the actual CMC is difficult to know. But it is safe to say that in real world applications, when used at 4 times the CMC, it is unlikely that you will be below the actual CMC.
If you know that your application only requires a slight amount of emulsifying and you are doing very light duty cleaning, in principle you can use approximately 1/4 the recommended dose for Alconox, LLC detergent and still be above the CMC. Of course the cleaning capacity of the detergent solution will be significantly reduced, so it is not usually recommended to use reduced concentrations of detergent. In very difficult to rinse applications such as filter cleaning, we often recommend a 1/2 -3/4 the recommended dose because this still gives you some excess concentration of detergent above the CMC, but leaves less detergent that needs to be rinsed away.
Note that most Alconox, LLC detergents are used at 1-3% for typical applications.
A micelle is an aggregate of surfactant molecules in a liquid solution. Micelles form only when the concentration of surfactant is greater than the (CMC), and the temperature of the system is sufficient for micelle formation. The utility of a micelle formation is that it can emulsify a contaminant that would otherwise be insoluble. This allows for it to be removed and the surface from which it came, can in turn be cleaned.
The CMC varies with electrolyte and residue content of solutions. It generally decreases as micelles shift from globes to rods in a more hexagonal arrangement in response to electrolyte content. The CMC of the individual surfactants is sometimes known at 25°C in DI water. For example, both Liquinox® Critical Cleaning Liquid Detergent and Citranox® Liquid Acid Cleaner and Detergent contain similar mixtures of anionic and nonionic surfactants that form mixed micelles. The mixed micelle CMC can be experimentally derived for any specific set of conditions. In general, a mixed micelle CMC will be lower than the average of the CMC of the constituent surfactants. Again, this mixed CMC value will be dependent on temperature and residual contamination.
To request an Alconox, LLC detergent for free, please complete the questionnaire at Get Sample. For more information about any one of our Alconox, LLC detergents, consult the technical bulletin for each product. Or click here to access each of our detergent’s Safety Data Sheets.
Do you have a critical cleaning question for the experts at Alconox, LLC? Search TechNotes to see if it’s been answered before or Ask Alconox.



