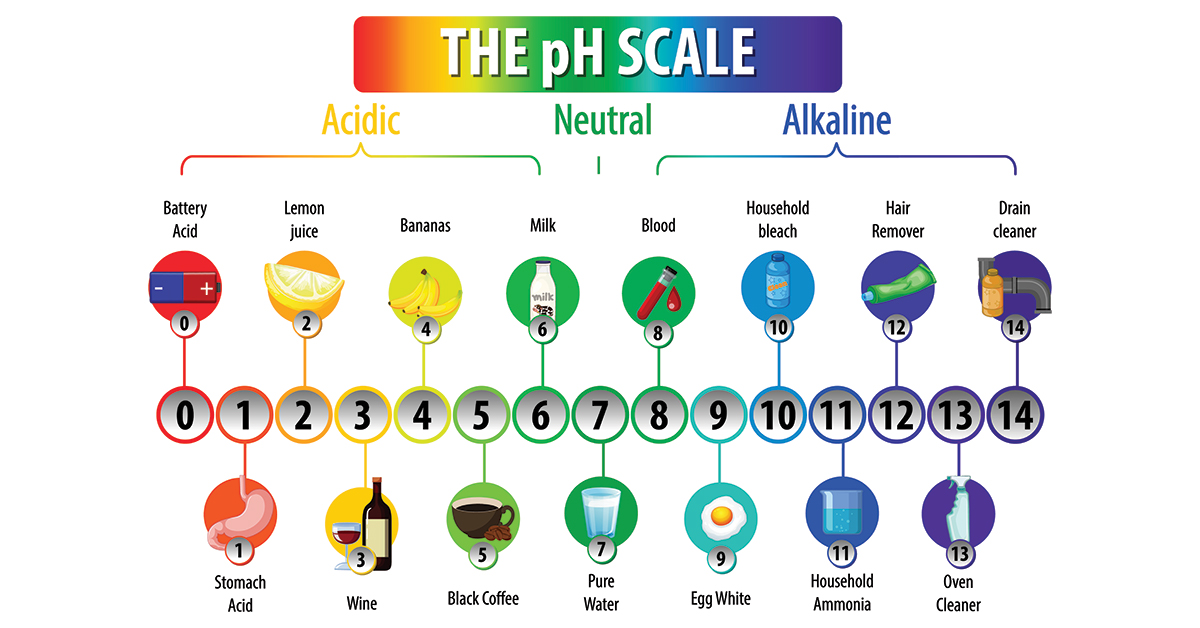
What is Halojet detergent?

Q: We run a mid-size contract manufacturing facility and have been struggling to clean our mixing tanks and filling equipment between production runs via CIP. Residues from our pigmented formulations, including foundations, waxes, tinted moisturizers, and similar products. A colleague mentioned new Halojet detergent at a recent industry event. Can you tell us more about it?……
Read more >