Specific Formulations for Cold Water Cleaning?
Q: I’m looking for detergents similar to Liquinox and Citranox, that are specially formulated for cold water cleaning conditions. Can you recommend analogs to the two?
A: For most residues, the detergency of an aqueous detergent is enhanced and hastened by heat.
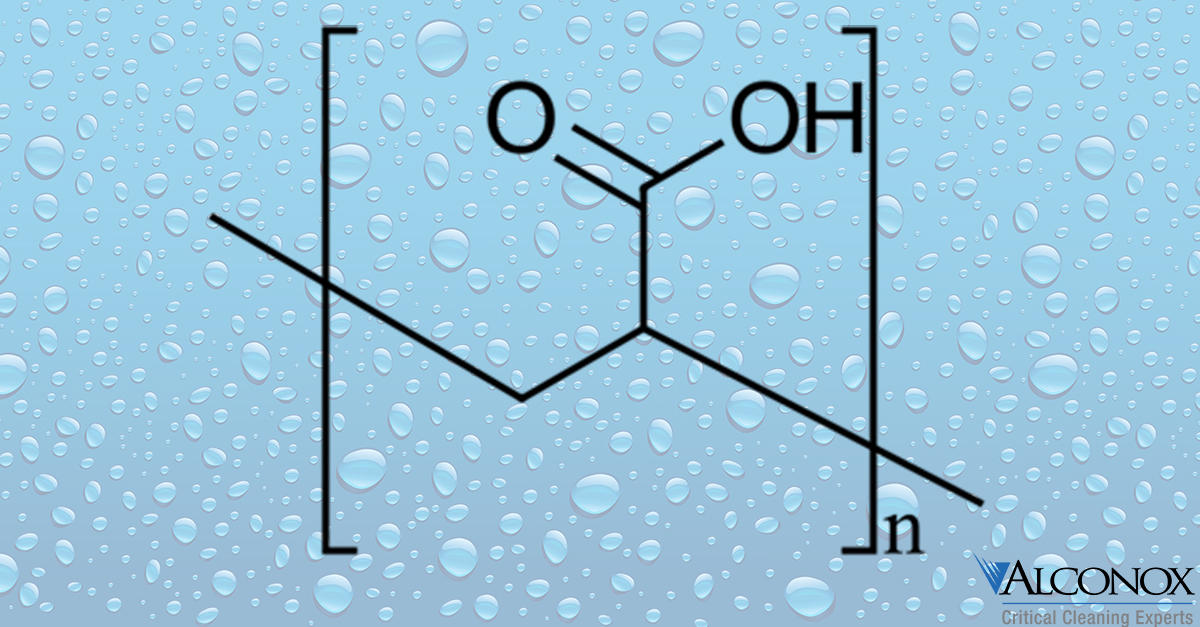
How to: Clean Carbomer
Q: We are having difficultly cleaning carbomer gel residue from our equipment. What do you recommend?
A: Carbomer or polyacrylic acid is a thickening agent, used to stabilize and provide the base for many ointments, cremes and lotions. As one might guess, it is a difficult residue. In the presence of water it can thicken and become a gelatinous, sticky gel. A goo.
Cleaning a Collagen Coating
Q: We are currently looking to remove a collagen coating from PET fabric and was wondering what we should use. We have Alconox powder, will that work?
A: To clean polyethylene terephthalate (PET) we generally want to stay away from alkaline and high alkaline detergents. Mildly alkaline is expected to be ok (< 10 pH). As with all compatibility matters....
Removing Isocyanate Residue
Q: Does your company sell any solutions for residual isocyanates? We currently use Detonox detergent. How long can it last?
A: In the past we have recommended the very same powerfully emulsifying Detonox® Ultimate Precision Cleaner you are currently using with great results.
Mystery Residue: Left Over Detergent Residue?
Q: Whenever the Detonox is left in a stainless steel sink, it forms a white substance at the bottom of the sink. We have tested the water with Detonox in a glass beaker and it does not form the same ‘Scum’.…